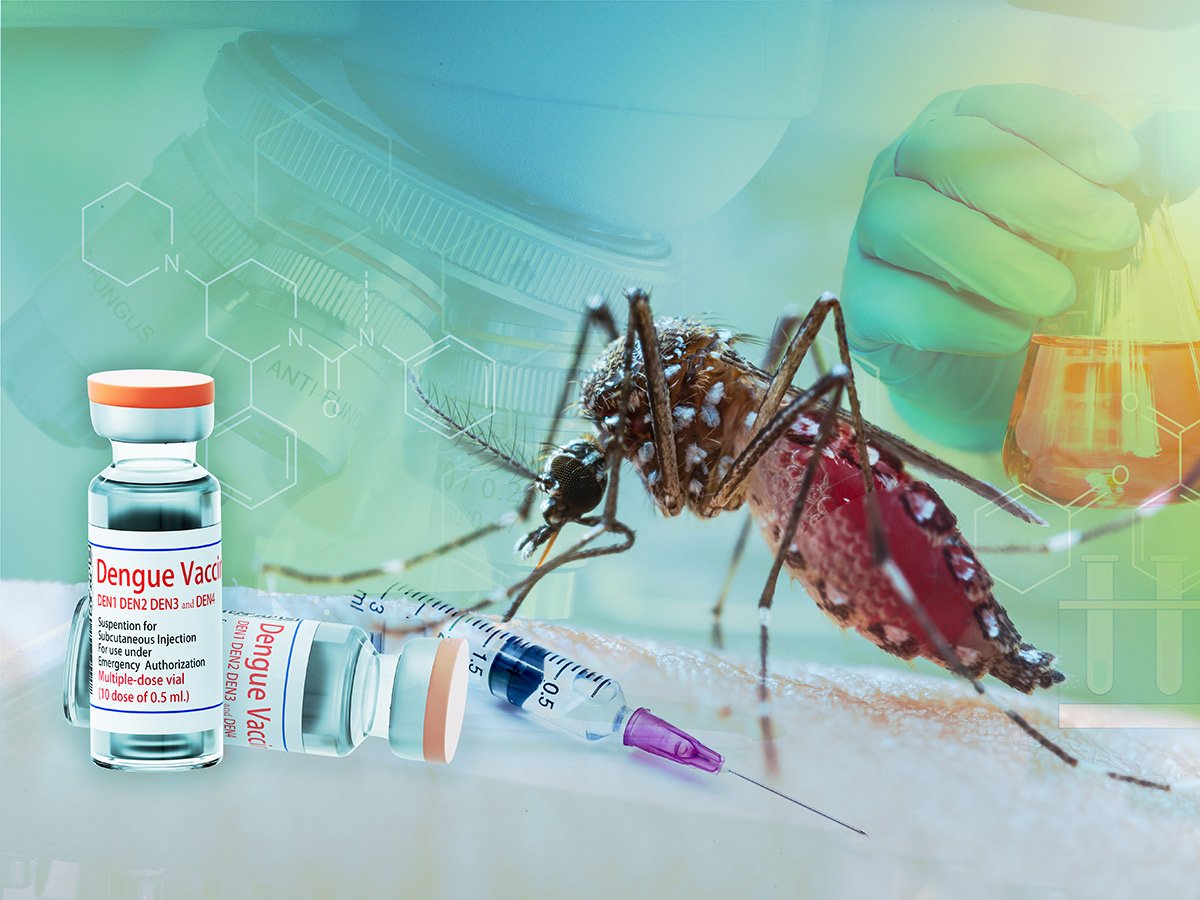
Pune: India has taken a big step in the prevention of dengue infection. The country’s first indigenous vaccine (Anti-Dengue Vaccine) developed from the virus strain found in America was successful in the initial trial, due to which the government has given permission for the next trial.
Pune-based Serum Institute of India (SII) told in the expert working committee meeting held last week that in the first trial, the vaccine (Anti-Dengue Vaccine) was tested on 60 people with placebo. After monitoring for more than six months, the vaccine was found to be completely safe in adults and effective on all four strains of the virus. After this, the Central Drugs Standard Control Organization (CDSCO) also gave approval for the second phase. In fact, the US National Institute of Allergy and Infectious Diseases (NIAID) provided isolated strains of dengue virus to SII, through which tetravalent live attenuated anti-dengue vaccine has been developed in India. The first trial was completed from 25 May 2019 to 29 December 2023, which also included patients infected with Den 1, Den 2, Den 3 and Den 4 strains of dengue.
Apart from SII in India, Indian Immunologicals Ltd. (IIL) is also working on the dengue vaccine (Anti-Dengue Vaccine). Recently, IIL also expressed hope to launch the vaccine commercially in early 2026. For this also, the US-based National Institute of Health (NIH) has provided the necessary virus.
This juice is effective in preventing dengue, prepare it like this

A senior member of the Expert Working Committee (SEC) said that two more tests are yet to be conducted on this indigenous vaccine (Anti-Dengue Vaccine), of which the second phase trial can be started in the next few months or weeks. This test will be done in big hospitals of the country, where the number of dengue infected patients will be sufficient. This may include medical institutions like Delhi AIIMS, PGI Chandigarh and CMC Vellore.
These are the results
The vaccine was found to be highly immunogenic in most of the participants involved in the first trial. During this time, some adverse effects have also come to the fore, which need to be discussed. These include erythema at the injection site, rash, headache, fatigue, muscle pain and joint pain. However, they have been completely resolved. Of the 60 participants (40 given the vaccine) and (20 given placebo), 23 participants (59%) started showing effect against all four strains of dengue between the ninth and 11th day, which was visible even after about 181 days.